Our commitment with quality is real and it’s present in everything we do on a daily basis.
We want to help our partners, customers and others stakeholders to improve all the quality related procedures across areas, optimizing their value chain with better outcomes, through our knowledge and experience leading to a operational efficiency and value-added products and results.
Our Quality team has intense training and experience in several fields which provides them a wider and strategical point to any case and project.
GENOTRIX, S.A. (or simply ‘GENOTRIX’) is a Portuguese company located at Trofa, which operates within the manufacture and distribution of medical devices and other innovative products. Its management values Quality as much more than a business requirement: it is regarded as a whole way of thinking and conducting all the activities that take place at the company and on behalf of the company.
GENOTRIX selects the most advanced technologies for designing, developing and manufacturing its products and medical devices. GENOTRIX always aims for applying the latest medical breakthroughs and strongly validates all products before release. Internal policy also comprises a permanent demand for the highest quality standards available to be pursued, implemented, followed and continuously improved at all times.
GENOTRIX fully complies with all local laws and regulations, as well as with all relevant and applicable international standards and agreements. At the same time, GENOTRIX management stands up for a flawless ethical behaviour, which aims for ensuring a constant fulfilling of the terms and requirements from both suppliers and customers, and also the complete protection of all data and confidentiality agreements.
Therefore, GENOTRIX is committed to:
• Identifying and applying good and efficient professional practices;
• Complying with applicable laws and regulations;
• Developing, applying, maintaining and continual improvement of effectiveness of Quality Management System according to EN ISO 9001 – Quality management and EN ISO 13485 – Quality management for the industry of medical devices;
• Ensuring patient safety through risk management, quality assurance of medical and In Vitro Diagnostics devices and compliance with Medical Device Regulation (MDR) 2017/745 and In Vitro Diagnostics Regulation 2017/746, respectively
• Ensuring the highest standards of device safety and performance;
• Obtaining the certification for both the referred quality management standards and European regulations;
• Continually improve processes and the quality of products and services.
In order to achieve these goals, GENOTRIX proposes to:
• Recruit, train, and support appropriately qualified staff;
• Appropriately evaluate customers’ (and potential customers’) needs and expectations;
• Acquire and ensure the efficient maintenance of state-of-the-art equipment;
• Identify and assess appropriate supplies and suppliers;
• Implement and follow verified and certified methods and processes;
• Validate all products and batches before release;
• Monitor key performance indicators.
Every single staff member, at every level of the company, is expected to demonstrate his or her own undeniable commitment to this Quality Policy.
Below you can have an overview of some of our QMS components plans:
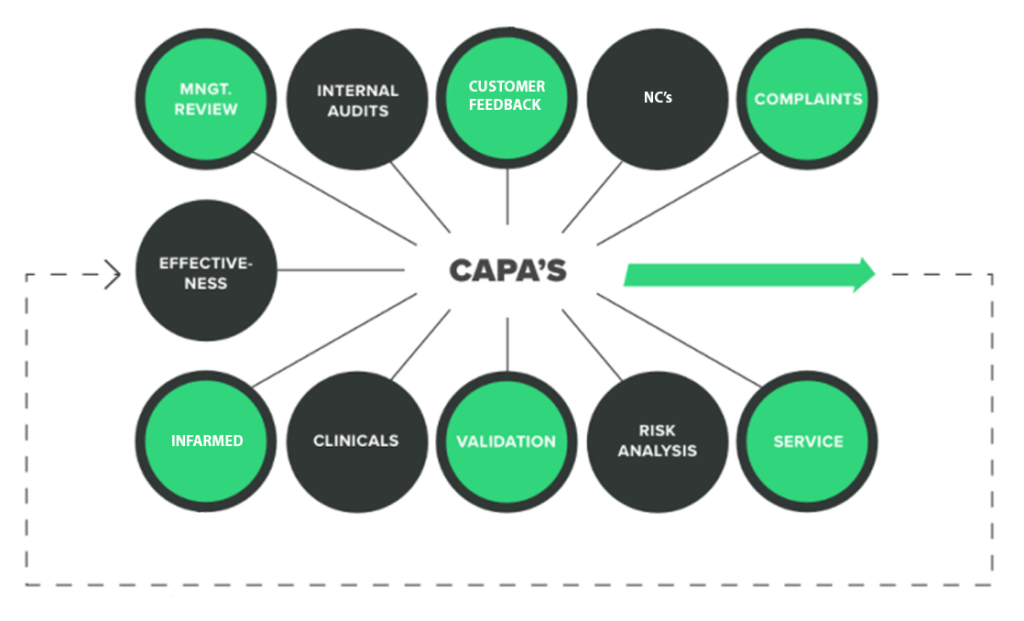
GENOTRIX QMS lifecycle requirements and role:
All the information is documented in our quality manual.
All the projects for Medical Devices have a QMS plan with all the specific requirements covering the strategic and regulatory specifications:
Our QMS plan will cover all aspects and lifecycle against the standards to evaluate and confirm the deliverables.

Lorem ipsum dolor sit amet, consectetur quam adipiscing quam elit tellus, luctus nec dapibus Vestibulum id ligula porta
Necessary cookies are absolutely essential for the website to function properly. These cookies ensure basic functionalities and security features of the website, anonymously.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
Functional cookies help to perform certain functionalities like sharing the content of the website on social media platforms, collect feedbacks, and other third-party features.
Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors.
Analytical cookies are used to understand how visitors interact with the website. These cookies help provide information on metrics the number of visitors, bounce rate, traffic source, etc.
Advertisement cookies are used to provide visitors with relevant ads and marketing campaigns. These cookies track visitors across websites and collect information to provide customized ads.
Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet.
Sign up to receive updates, promotions, and sneak peaks of upcoming products. Plus 20% off your next order.